Blog
Vaccine Storage Is A Key To Effective Prevention

Barbara Graber, manager of compliance and special projects at LaSalle says “We’ve taken every step necessary to insure vaccines are received, handled and stored properly so as not to compromise their efficiency as well as protect the families we serve.”
The CDC and state VFC programs reiterated their recommendations to healthcare providers on best practices in storing and handling vaccines.
(San Bernardino, CA) Each year millions of dollars are lost in the United States because healthcare providers improperly store and handle vaccines meant to protect against childhood diseases such as measles and chicken pox.
LaSalle Medical Associates, Inc. wants to assure parents, and the community, it follows the Centers for Disease Control and Prevention and Vaccine for Children programs’ vaccine guidelines.
“This is something we take very seriously. Our vaccine handling and storage techniques are audited annually by either the CHDP or VFC,’’ says Barbara Graber, manager of compliance and special projects at LaSalle. “We’ve taken every step necessary to insure vaccines are received, handled and stored properly so as not to compromise their efficiency as well as protect the families we serve.”
Just last June and July, VFC and CHDP representatives audited LaSalle’s vaccine storage and handling practices, Graber says. And, two years ago LaSalle purchased, and placed, Pharmaceutical-grade refrigerators in each of its four clinics. Those refrigerators are exclusively for vaccine storage and, the temperatures in each refrigerator are monitored twice daily and logged, Graber says.
“According to regulations we keep these logs for three years and also have protocols in place in the event of an emergency or power outage,’’ Graber says.
Recently, the CDC and state VFC programs reiterated their recommendations to healthcare providers on best practices in handling vaccines. Both recommend instituting vaccine plans. This plan should include routine vaccine management – from ordering and controlling inventory to storing and monitoring storage conditions, according to the CDC.
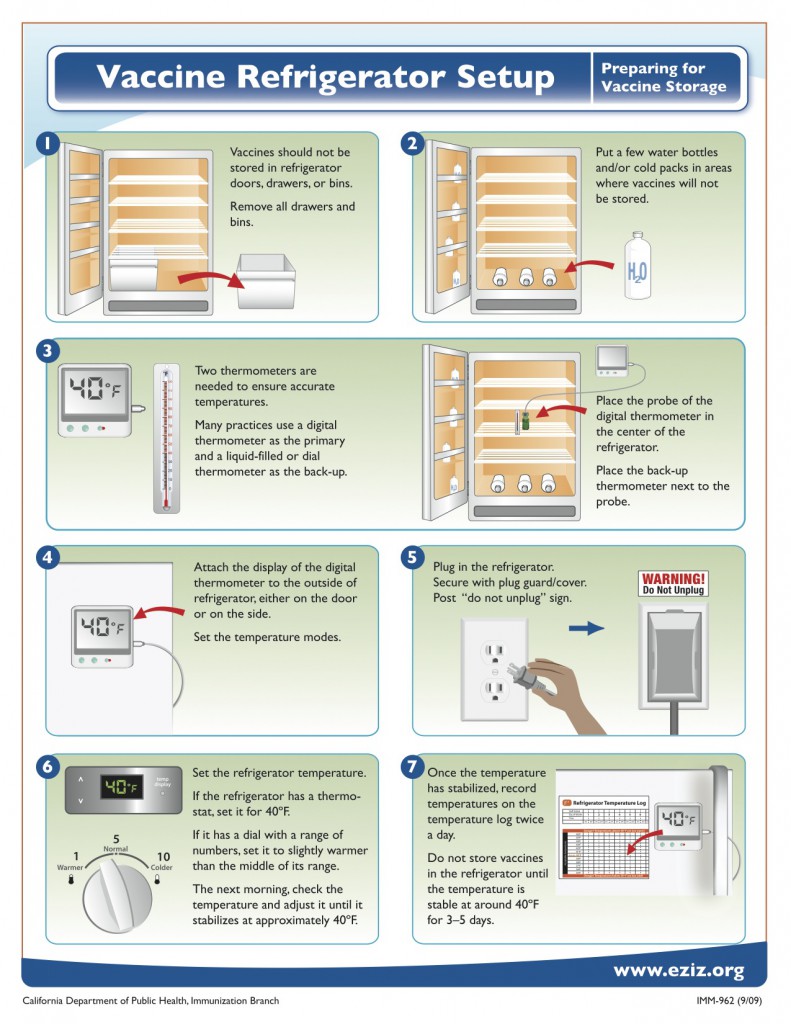
The CDC also recommends healthcare providers store vaccines in refrigerators without freezers. However, a combination refrigerator/freezer unit sold for home use can be used for vaccine storage if the refrigerator and freezer compartments each have separate external doors. Refrigerators must have a calibrated thermometer in place, and checked twice daily for temperature compliance.
It is also recommended the plan include protocols for emergency vaccine retrieval and storage in the event of a refrigerator or freezer malfunction, power failures, or other emergencies which might compromise vaccine storage conditions.
To minimize loss, the CDC recommends storage and handling plans to include:
- Up to date contact information.
- Descriptions of the roles and responsibilities of primary and back up vaccine coordinators.
- Summaries of storage requirements for each vaccine in inventory.
- Protocols for vaccine storage unit temperature monitoring, storage equipment maintenance, placement of vaccine within storage units, responding to vaccine storage and handling problems, transporting and receiving vaccine shipments, and proper disposal of vaccines and supplies.
The CDC also recommends healthcare providers store vaccines in refrigerators without freezers. However, a combination refrigerator/freezer unit sold for home use can be used for vaccine storage if the refrigerator and freezer compartments each have separate external doors. Refrigerators must have a calibrated thermometer in place, and checked twice daily for temperature compliance.
Graber says LaSalle has, and will continue to, meet all of the CDC vaccine recommendations. In addition, LaSalle has its own internal policies and procedures which coincide with federal and state recommendations, she says.
“This is both a healthcare and fiscal issue,’’ says LaSalle Executive Vice President Carl Maier. “We are diligent in protecting the patients we serve as well as mindful of costs, which is a reason we allocated the time and funding purchasing high quality refrigeration equipment.”
For more information call LaSalle Medical Associates, Incorporated at (909) 890-0407.
About LaSalle Medical Associates
LaSalle Medical Associates, Inc., operates four clinics employing more than 120 dedicated healthcare professionals, treating children, adults, and seniors in San Bernardino County. LaSalle’s patients are primarily served by Medi-Cal and they also accept Blue Cross, Blue Shield, Molina, Care 1st, Health Net and Inland Empire Health Plan (IEHP). LaSalle’s clinics are at 17577 Arrow Blvd. in Fontana, 16455 Main St. in Hesperia and 1505 West 17th St. and 565 N. Mt. Vernon Ave. in San Bernardino.
LaSalle Medical Associates, Inc., is also an Independent Practice Association (IPA) of independently contracted doctors, hospitals and clinics, delivering high quality care to more than 245,000 patients in Fresno, Kings, Los Angeles, Madera, Riverside, San Bernardino, San Joaquin and Tulare Counties.
LaSalle’s IPA members in the Inland Empire include: LaSalle Medical Associates, Arrowhead Orthopedics, Arther Medical Group, Rancho Medical Group, Rancho Paseo Medical Group, Alessandro Medical Group Banning Medical Group, and San Bernardino Urological Associates. Hospital affiliations include: Rancho Springs Medical Center, Arrowhead Regional Medical Center, Community Hospital of San Bernardino, St. Bernadine Medical Center, Mountains Community Hospital, Redland Community Hospital, Loma Linda University Medical Center, Corona Regional Medical Center, Riverside County Medical Center, Parkview Community Hospital.
-end-
